Research focus
Our research activities are focused on disease modeling using induced pluripotent stem cells (iPSC) from patients and their CRISPR-Cas9 gene edited controls. We have a specific disease-oriented focus on Alzheimer’s disease (AD) Frontotemporal dementia (FTD), Schizophrenia, Epilepsy and Neurofibromatosis 1 (NF1).
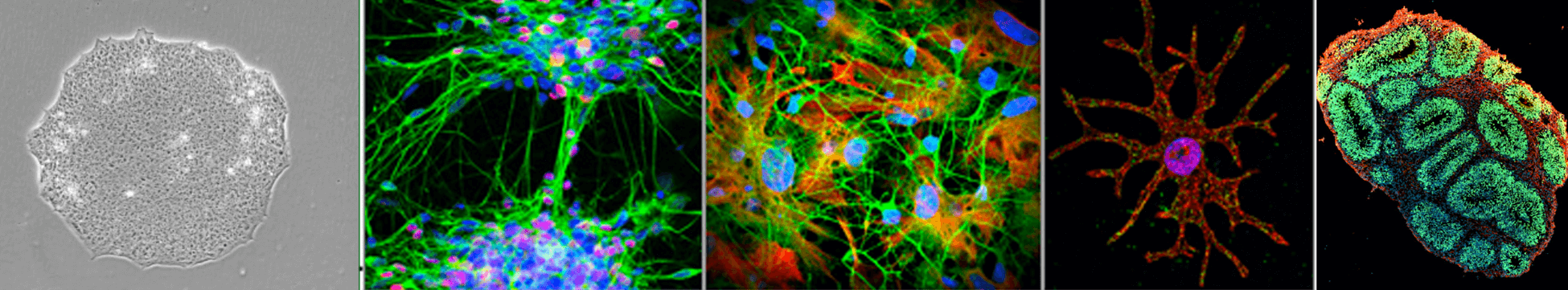 Within these diseases we are investigating cell type specific pathologies in neurons, astrocytes and microglia. We are conducting those studies in 2D and in 3D organoid structure. We are especially keen on understanding overlapping dysfunctions amongst different types of neurodegenerative and neurodevelopmental disorders and identification of key players in disease pathways. We believe that comprehensive understanding of such molecular commonalities may provide opportunities for novel interventions that are beneficial for an array of related diseases facilitating cost effective drug development. Furthermore, our lab is working in early embryology and epigenetic alterations leading to developmental abnormalities and infertility.
Within these diseases we are investigating cell type specific pathologies in neurons, astrocytes and microglia. We are conducting those studies in 2D and in 3D organoid structure. We are especially keen on understanding overlapping dysfunctions amongst different types of neurodegenerative and neurodevelopmental disorders and identification of key players in disease pathways. We believe that comprehensive understanding of such molecular commonalities may provide opportunities for novel interventions that are beneficial for an array of related diseases facilitating cost effective drug development. Furthermore, our lab is working in early embryology and epigenetic alterations leading to developmental abnormalities and infertility.
Current projects
Our primary research focus is on Alzheimer's disease (AD), with particular emphasis on understanding why women are at increased risk of developing the disease. While amyloid plaques and tau tangles are hallmark pathologies, growing evidence highlights neuroinflammation as a central driver of AD progression. Large-scale genetic studies have identified numerous AD-associated risk variants in immune-related genes, many of which are highly expressed in microglia, the brain’s resident immune cells.
We investigate how sex-specific genetic and epigenetic mechanisms shape microglial function and contribute to the higher vulnerability of women to AD. A key focus is the role of immune-related genes on the X chromosome, including age-related reactivation of genes on the inactive X chromosome, and how these processes influence inflammatory responses in female microglia (funded by the Lundbeck Foundation, Ascending Investigators). This project is in collaboration with Mathew Blurton-Jones (UC Irvine, USA) and Jan Żylicz, ReNEW.
In idiopathic AD, we study the role of RhoA and genetic risk single nucleotide polymorphisms in iPSC-derived neurons, astrocytes, and microglia (RhoAD, funded by the Novo Nordisk Foundation). This project is in collaboration with Morten Meyer at SDU.
Through the DEVELOPNOID project (funded by the Lundbeck Foundation), we examine post-translational modifications (PTMs) and early alterations in synaptic pruning using cerebral organoids derived from familial AD patient iPSCs, their isogenic controls, and idiopathic AD patient iPSCs. This collaborative project involves Martin Røssel-Larsen at SDU and Madeline Lancaster at University of Cambridge. Within DEVELOPNOID, we also investigate the role of miRNAs in Schizophrenia, integrating miRNA-seq, RNA-seq, and proteomics to identify converging disease pathways and regulatory hubs in collaboration with Michael Benros, Rigshospital, Martin Røssel Larsen, SDU and Boye Schnack Nielsen, Bioneer A/S.
Our laboratory actively contributes to Neuroscience Academy Denmark, where I supervise PhD students conducting research projects in epilepsy (role of extra synaptic GABA receptors in epilepsy, Jonas Hald) , Alzheimer's disease (Alterations in microglial EV composition and content in idiopathic AD, Rita Bessa) and Neurofibromatosis type 1 (NF1)(Brain organoid models to understand cognitive dysfunction in NF1, Angelika Langeder). The epilepsy project is in close collaboration with Jakob Baslev Sørensen, UCPH and Rikke Møller, Filadelfia. The AD project is in collaboration with Morten Blaabjerg, SDU and the NF1 project is in close collaboration with Rene Mathiasen and Stense Farholt, Rigshospital as well as Sara Brebbia Dirksen NF Denmark.
Within veterinary sciences, we generate and apply animal iPSCs. We have successfully established porcine iPSCs and canine iPSC-like cells. Our canine iPSC work focuses on dogs with canine cognitive dysfunction (CCD) as a natural model of human AD (funded by the DFF). Porcine iPSCs have been implemented to develop non-immunogenic iPSC platforms in collaboration with LifePharm and the Novo Nordisk Stem Cell Department.
Currently, we are developing bovine iPSCs to investigate the safety of CRISPR-based gene editing in livestock (CRISPRcow, funded by DFF) collaboration with Fabianna Bressan, University São Paulo, and generating equine iPSCs to establish immune-evasive treatment strategies for osteoarthritis in horses (StaySTEM, funded by DFF), collaboration with Per Hölmich and Jasmin Bagge, Hvidovre Hospital and Bjørn Holst, Bioneer A/S.
- Stoklund Dittlau, K, Terrie, L, Baatsen, P, Kerstens, A, De Swert, L, Janky, R, Corthout, N, Masrori, P, Van Damme, P, Hyttel, P, Meyer, M, Thorrez, L, Freude, K. & Van Den Bosch, L. FUS-ALS hiPSC-derived astrocytes impair human motor units through both gain-of-toxicity and loss-of-support mechanisms. Molecular Neurodegeneration. 2023. DOI: https://doi.org/10.1186/s13024-022-00591-3
- Corsi, G, Gadekar, V, Haukedal, H, Doncheva, N, Anthon, C, Ambardar, S, Palakodeti, D, Hyttel, P, Freude, K, Seemann, S & Gorodkin, J. The transcriptomic landscape of neurons carrying PSEN1 mutations reveals changes in extracellular matrix components and non-coding gene expression. Neurobiology of Disease. 2023. DOI: https://doi.org/10.1016/j.nbd.2022.105980
- Chandrasekaran, A, Dittlau, K, Corsi, G, Haukedal, H, Doncheva, N, Ramakrishna, S, Ambardar, S, Salcedo, C, Schmidt, SI, Zhang, Y, Cirera, S, Pihl, M, Schmid, B, Nielsen, T, Nielsen, J, Kolko, M, Kobolak, J, Dinnyes, A, Hyttel, P, Palakodeti, D, Gorodkin, J, Muddashetty, R, Meyer, M, Aldana B, Freude K. 2021. Astrocytic reactivity triggered by defective autophagy and metabolic failure activates NF-kB signaling and causes neurotoxicity in frontotemporal dementia type 3. Stem Cell Reports. 2021. DOI: https://pubmed.ncbi.nlm.nih.gov/34678206/
- Andersen, JV, Skotte, NH, Christensen, SK, Polli, FS, Shabani, M, Markussen, KH, Haukedal, H, Westi, EW, Diaz-delCastillo, M, Sun, RC, Kohlmeier, KA, Schousboe, A, Gentry, MS, Tanila, H, Freude, K, Aldana, BI, Mann, M & Waagepetersen, HS, Hippocampal disruptions of synaptic and astrocyte metabolism are primary events of early amyloid pathology in the 5xFAD mouse model of Alzheimer's disease, Cell Death & Disease. 2021. DOI: https://doi.org/10.1038/s41419-021-04237-y
- Ramakrishna S, Jhaveri V, Konings S, Chakraborty S, Holst B, Schmid B, Freude K, Gouras G, Muddashetty R. APOE4 affects basal and NMDAR mediated protein synthesis in neurons by perturbing calcium homeostasis. Journal of Neuroscience. 2021. DOI: https://doi.org/10.1523/JNEUROSCI.0435-21.2021
- Chandrasekaran A, Jensen P, Mohamed F, Lancaster M, Benros M, Larsen M, Freude K. A protein-centric view of in vitro biological model systems for Schizophrenia. Stem Cells. 2021. https://doi.org/10.1002/stem.3447.
- Byman, E, Martinsson, I, Haukedal, H, Netherlands Brain, B, Gouras, G, Freude, K, & Wennstrom, M. Neuronal alpha-amylase is important for neuronal activity and glycogenolysis and reduces in presence of amyloid beta pathology. Aging Cell. 2021. DOI: https://doi.org/10.1111/acel.13433
- Schmidt SI, Bogetofte H, Ritter L, Agergaard JB, Hammerich D, Kabiljagic AA, Wlodarczyk A, Lopez SG, Sørensen MD, Jørgensen ML, Okarmus J, Serrano AM, Kristensen BW, Freude K, Owens T, Meyer M. Microglia-Secreted Factors Enhance Dopaminergic Differentiation of Tissue- and iPSC-Derived Human Neural Stem Cells. Stem Cell Reports. 2021. DOI: https://doi.org/10.1016/j.stemcr.2020.12.011
- Frederiksen HR, Doehn U, Tveden-Nyborg P, Freude K. Non-immunogenic Induced Pluripotent Stem Cells, a Promising Way Forward for Allogenic Transplantations for Neurological Disorders. Frontiers in Genome Editing. 2020. DOI: https://doi.org/10.3389/fgeed.2020.623717
- Aldana BI, Zhang Y, Jensen P, Chandrasekaran A, Christensen SK, Nielsen TT, Nielsen JE, Hyttel P, Larsen MR, Waagepetersen HS, Freude K. Glutamate-glutamine homeostasis is perturbed in neurons and astrocytes derived from patient iPSC models of frontotemporal dementia. Mol Brain. 2020. DOI: https://doi.org/10.1186/s13041-020-00658-6
- Haukedal H, Freude K. Implications of Microglia in Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. J Mol Biol. 2019.DOI: https://doi.org/10.1016/j.jmb.2019.02.004
- Vohra R, Aldana BI, Skytt DM, Freude K, Waagepetersen H, Bergersen LH, Kolko M. (2018). Essential Roles of Lactate in Muller Cell Survival and Function. Mol Neurobiol. 2018. DOI: https://doi.org/10.1007/s12035-018-1056-2
- Ochalek A, Mihalik B, Avci HX, Chandrasekaran A, Teglasi A, Bock I, Giudice ML, Tancos Z, Molnar K, Laszlo L, Nielsen JE, Holst B, Freude K, Hyttel P, Kobolak J, Dinnyes A. Neurons derived from sporadic Alzheimer's disease iPSCs reveal elevated TAU hyperphosphorylation, increased amyloid levels, and GSK3B activation. Alzheimers Res Ther. 2017. DOI: https://doi.org/10.1186/s13195-017-0317-z
- Zhang, Y, Schmid, B, Nikolaisen, NK, Rasmussen, MA, Aldana, BI, Agger, M, Calloe, K, Stummann, TC, Larsen, HM, Nielsen, TT, Huang, J, Xu, F, Liu, X, Bolund, L, Meyer, M, Bak, LK, Waagepetersen, HS, Luo, Y, Nielsen, JE, Consortium, FR, Holst, B, Clausen, C, Hyttel, P, Freude, K. Patient iPSC-Derived Neurons for Disease Modeling of Frontotemporal Dementia with Mutation in CHMP2B. Stem Cell Reports . 2017. DOI: https://doi.org/10.1016/j.stemcr.2017.01.012
- Freude, K., Penjwini, M., Davis, J.L., LaFerla, F.M., Blurton-Jones, M., Soluble amyloid precursor protein induces rapid neural differentiation of human embryonic stem cells. J Biol Chem . 2011. DOI: https://doi.org/10.1074/jbc.M111.227421
- Kalscheuer, VM, Freude, K, Musante, L, Jensen, LR, Yntema, HG, Gecz, J, Sefiani, A, Hoffmann, K, Moser, B, Haas, S, Gurok, U, Haesler, S, Aranda, B, Nshedjan, A, Tzschach, A, Hartmann, N, Roloff, TC, Shoichet, S, Hagens, O, Tao, J, Van Bokhoven, H, Turner, G, Chelly, J, Moraine, C, Fryns, JP, Nuber, U, Hoeltzenbein, M, Scharff, C, Scherthan, H, Lenzner, S, Hamel, BC, Schweiger, S, Ropers, HH. Mutations in the polyglutamine binding protein 1 gene cause X-linked mental retardation. Nat Genet. 2003. DOI: https://doi.org/10.1038/ng1264
Click on the name below to see a complete overview of the senior group members' publications:
Research Funding
 |
 |
 |
 |
Group Leader
Professor Kristine Freude, PhD
Grønnegårdsvej 7
DK-1870 Frederiksberg C
Phone: +45 25572261
Group members
|
Name |
Position |
| Afonso, Inês Rodrigues | Master student |
| Aldana, Blanca Irene | Associate Professor |
| Andersen, Sofie Amalie Flintholm | PhD student |
| Beckvard, Veronica | NAD rotation student |
| Bessa, Rita Goncalves | PhD NAD |
| Frederiksen, Henriette | Postdoc |
| Glahder, Trine Marie Ahlman | TAP |
| Hald, Jonas Laugård | PhD (NAD) |
| Jensen, Betina Wingreen | Project Manager |
| Jensen, Rikke Rejnholdt | Visiting PhD student |
| Jensen, Lars Jørn | Associate Professor |
| Kadlecova, Marion | PhD |
| Kovalova, Vera | Visiting Postdoc |
| Langeder, Angelika Birgit | PhD NAD |
| Mohamed, Fadumo | Guest researcher |
| Oskarsson, Chichi N. | TAP |
| Rasmussen, Laura | visiting PhD studen from SDU |
| Tao, Ruixin | PhD |
| Wathikthinnakon, Methi | Postdoc |
| Woodruff, Sophie Claire Bastrup | NAD rotation student |
| Yoon, Ye Eun | Visiting Postdoc |
